Prepared by Mousumi Islam
Entrepreneur | Business Leader | Policy Advocate | Global Connector | Champion of Women’s Empowerment | Social Visionary | Maritime & Blue Economy Leader | Healthcare & Medical Devices Industry Leader
Executive Summary
This fact sheet and research paper outline strategic innovation locations for the manufacturing of medical equipment and devices in Bangladesh, situating the country as a competitive hub within South Asia. The analysis identifies key zones, their comparative advantages, product specialization potential, export readiness, and partnership frameworks. The aim is to provide a national-level roadmap to policymakers, investors, and stakeholders.
Global Benchmarks of Innovation Hubs
- USA (Silicon Valley, Boston, Minnesota): AI-driven medical devices, surgical robotics, wearables.
- Europe (Germany, Switzerland, Ireland): Precision engineering, orthopedics, implants, CE-regulated production.
- Japan & South Korea: Imaging, miniaturization, smart hospital automation devices.
- China & Singapore: Large-scale, cost-effective manufacturing, telemedicine hardware, supply chain hubs.
Bangladesh Innovation & Manufacturing Zones
Dhaka North & South (Tejgaon, Uttara, Mirpur): Prototyping, patient monitors, OT lights/tables, sterilization units.
Gazipur Industrial Corridor: Mass manufacturing of consumables: syringes, IV sets, PPE, hospital furniture.
Narayanganj & Rupganj: Non-woven, disposables, sterile packaging solutions.
Kaliakoir Hi-Tech Park: Digital health integration: telemedicine kits, wearables, portable ultrasound.
Chattogram & EPZ: Export assembly hubs: dialysis modules, large equipment, logistics.
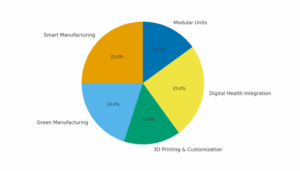
Innovation Priorities for Manufacturers
- Smart Manufacturing: IoT-enabled production, automation, AI quality control.
- Green Manufacturing: Renewable-powered facilities, biodegradable plastics, low-waste processes.
- 3D Printing & Customization: Personalized implants, prosthetics, orthopedic solutions.
- Digital Health Integration: Remote monitoring devices, cloud-linked ECG/IVD tools.
- Modular Units: Rapidly scalable decentralized factories, adaptable to demand.
Export Corridors
- Sea: Chattogram (primary), Mongla (secondary).
- Air: HSIA Dhaka for high-value, low-weight exports.
- Land: Benapole, Tamabil, Banglabandha for India and SAARC trade.
Partnerships & Testbeds
- Universities: BUET, MIST, Dhaka University, icddr, b for R&D and testing.
- Hospitals: BSMMU, DMCH, and private hospitals for clinical validation.
- International: JETRO/JICA, Enterprise Singapore, EU Notified Bodies, FDA consultants.
- Industry Associations: MEDMEB, BASIS, BGMEA for cross-industry synergies.
Policy & Incentives Framework
- ISO 13485 certification support for factories.
- Export cash incentives tied to CE/FDA pathways.
- Public procurement preference for domestic manufacturers.
- VAT rebates, bonded warehouses, and duty drawback schemes.
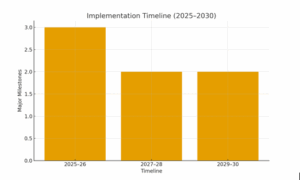
Implementation Timeline
- 2025–2026: Establish cleanrooms, pilot exports to SAARC, and DGDA approvals for 10 SKUs.
- 2027–2028: CE marking for priority devices, expansion into EU/GCC markets.
- 2029–2030: FDA submissions, turnkey clinic modules, global partnerships expansion.
Key Performance Indicators
- 10 ISO 13485 certified plants by 2030.
- 80 DGDA-approved SKUs by 2030.
- Export revenue target: USD 80 million by 2030.
- Creation of 3,000 direct jobs with 40% women participation.
Conclusion
Bangladesh has the potential to become South Asia’s hub for affordable, high-quality medical device manufa-cturing. By strategically positioning innovation zones, aligning with global best practices, and ensuring regulatory readiness, the nation can build a sustainable industry that serves both domestic healthcare needs and international markets.
Impact of the Fact Sheet
Policy-Level Impact
- Government Adoption: Provides a ready-made roadmap for DGDA, BIDA, and ministries to zone, incentivize, and regulate.
- Legitimacy for Advocacy: Positions Mousumi Islam as a policy advocate and industry pioneer.
- Evidence-Based Push: Supplies concrete benchmarks, zones, and data to accelerate regulatory reforms.
Business & Investor Impact
- Attracting FDI: Foreign investors gain confidence from mapped industrial clusters.
- Bank/DFI Confidence: Multilaterals and banks can justify financing cleanrooms, test labs, and sterilization plants.
- Export Deals: Creates credibility for long-term export partnerships with hospitals and distributors.
International Visibility Impact
- Positioning Bangladesh: Establishes Bangladesh as the ‘Affordable Innovation & Manufacturing Hub for South Asia.’
- Networking Edge: Acts as a conversation starter with JETRO, EU Horizon, Lloyd’s Register Foundation, WHO, and others.
Social & Economic Impact
- Healthcare Access: Lower device costs and faster access for domestic hospitals and clinics.
- Jobs & Skills: Over 3,000 direct jobs by 2030, with 40% women’s participation.
- Sustainability: Promotes green manufacturing aligned with UN SDGs.
In sum, this fact sheet is a national advocacy tool, an investor pitch, and a branding document that strengthens Bangladesh’s role in global medical devices supply chains.